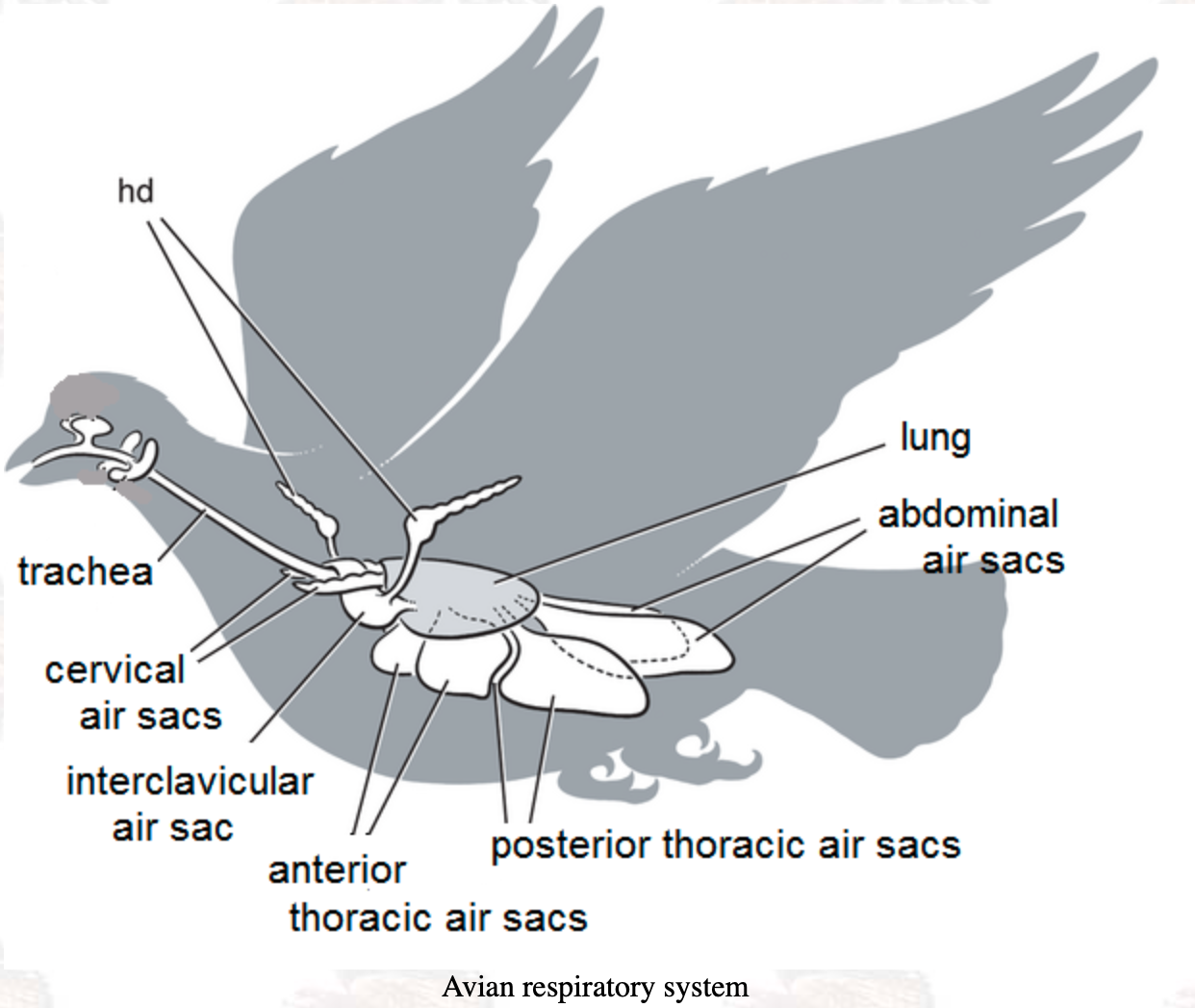
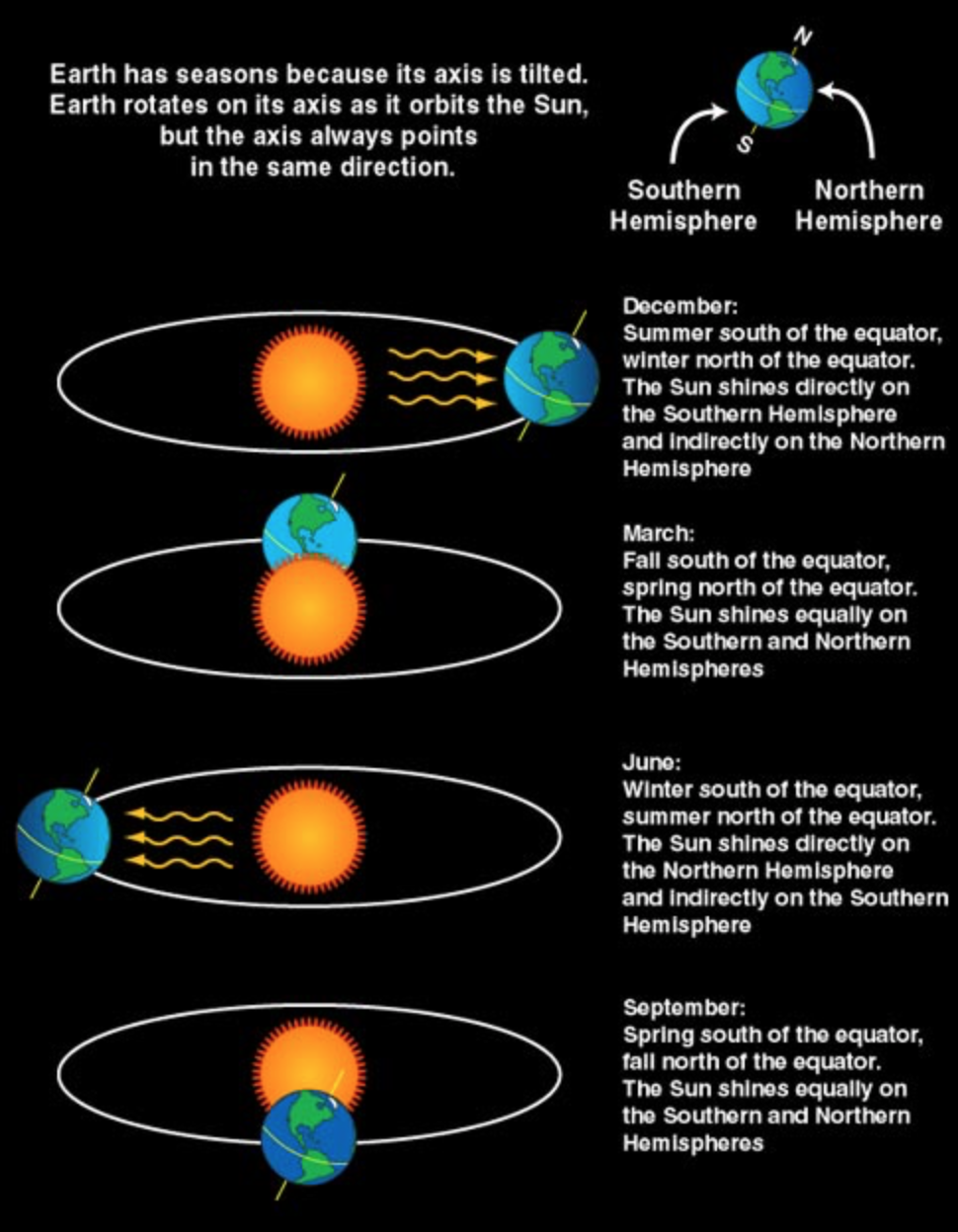
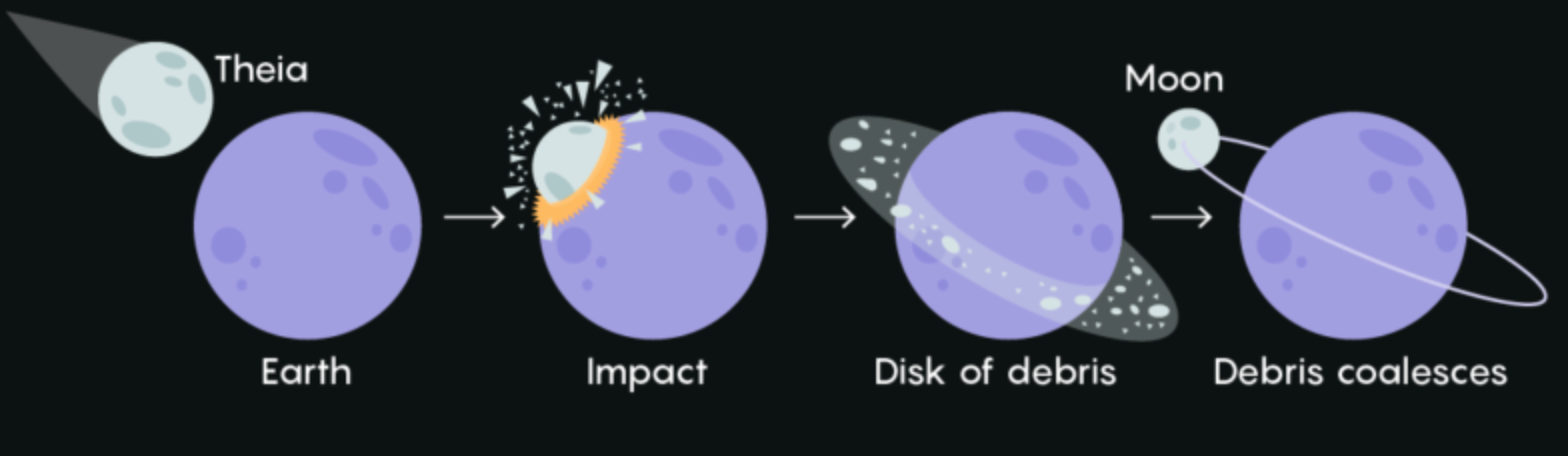
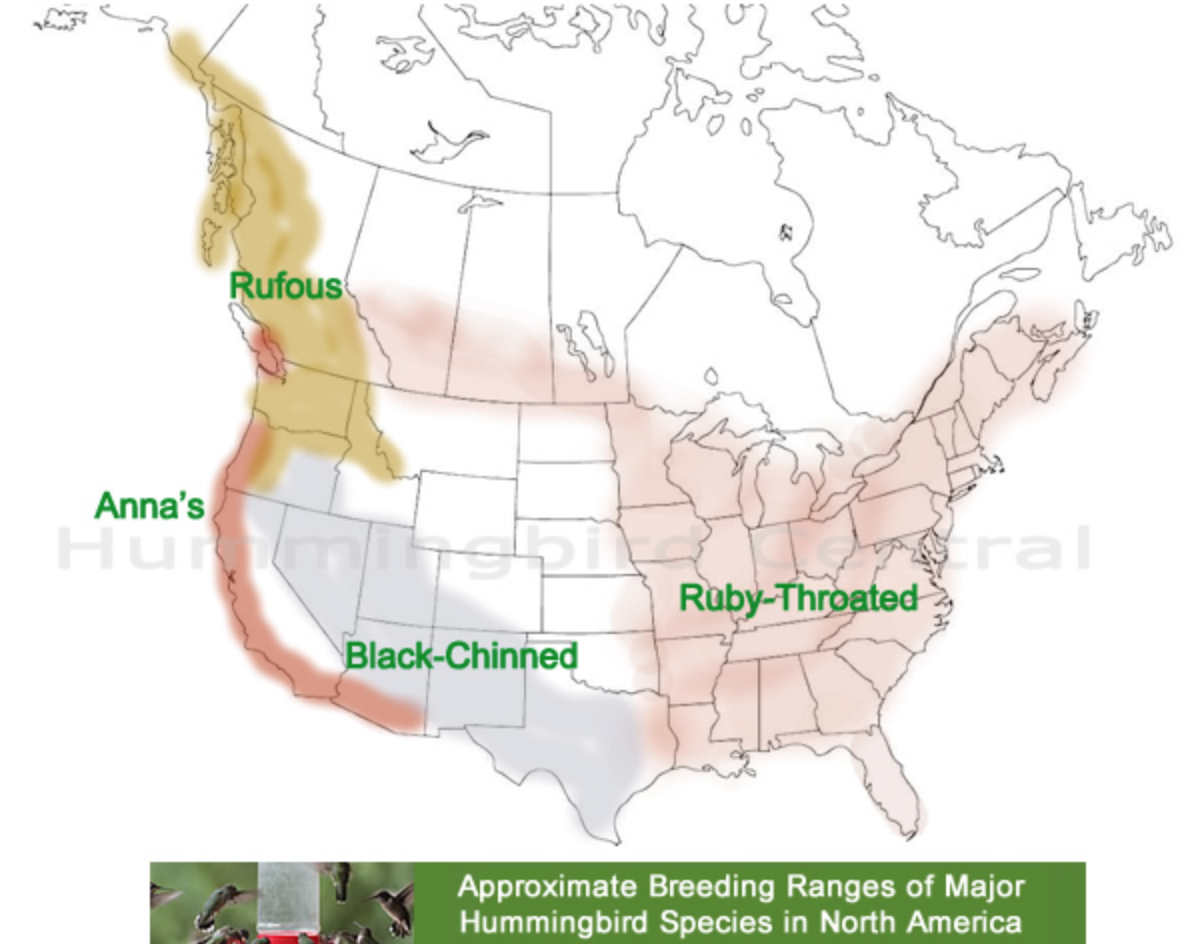
book info
excerpts:
First Part of the Ingenious Gentleman Don Quixote of La Mancha
Chapter I
Which describes the condition and profession of the famous gentleman Don Quixote of La Mancha
Somewhere in La Mancha, in a place whose name I do not care to remember, a gentleman lived not long ago, one of those who has a lance and ancient shield on a shelf and keeps a skinny nag and a greyhound for racing. An occasional stew, beef more often than lamb, hash most nights, eggs and abstinence on Saturdays, lentils on Fridays, sometimes squab as a treat on Sundays—these consumed three-fourths of his income. The rest went for a light woolen tunic and velvet breeches and hose of the same material for feast days, while weekdays were honored with dun-colored coarse cloth. He had a housekeeper past forty, a niece not yet twenty, and a man-of-all-work who did everything from saddling the horse to pruning the trees. Our gentleman was approximately fifty years old; his complexion was weathered, his flesh scrawny, his face gaunt, and he was a very early riser and a great lover of the hunt. Some claim that his family name was Quixada, or Quexada, for there is a certain amount of disagreement among the authors who write of this matter, although reliable conjecture seems to indicate that his name was Quexana. But this does not matter very much to our story; in its telling there is absolutely no deviation from the truth. And so, let it be said that this aforementioned gentleman spent his times of leisure—which meant most of the year—reading books of chivalry with so much devotion and enthusiasm that he forgot almost completely about the hunt and even about the administration of his estate; and in his rash curiosity and folly he went so far as to sell acres of arable land in very early riser and a great lover of the hunt. Some claim that his family name was Quixada, or Quexada, for there is a certain amount of disagreement among the authors who write of this matter, although reliable conjecture seems to indicate that his name was Quexana. But this does not matter very much to our story; in its telling there is absolutely no deviation from the truth.
And so, let it be said that this aforementioned gentleman spent his times of leisure—which meant most of the year—reading books of chivalry with so much devotion and enthusiasm that he forgot almost completely about the hunt and even about the administration of his estate; and in his rash curiosity and folly he went so far as to sell acres of arable land in order to buy books of chivalry to read, and he brought as many of them as he could into his house; and he thought none was as fine as those composed by the worthy Feliciano de Silva, because the clarity of his prose and complexity of his language seemed to him more valuable than pearls, in particular when he read the declarations and missives of love, where he would often find written: The reason for the unreason to which my reason turns so weakens my reason that with reason I complain of thy beauty. And also when he read: . . . the heavens on high divinely heighten thy divinity with the stars and make thee deserving of the deserts thy greatness deserves.
With these words and phrases the poor gentleman lost his mind, and he spent sleepless nights trying to understand them and extract their meaning, which Aristotle himself, if he came back to life for only that purpose, would not have been able to decipher or understand.
…
“O Princess Dulcinea, mistress of this captive heart! Thou hast done me grievous harm in bidding me farewell and reproving me with the harsh affliction of commanding that I not appear before thy sublime beauty. May it please thee, Señora, to recall this thy subject heart, which suffers countless trials for the sake of thy love.”
He strung these together with other foolish remarks, all in the manner his books had taught him and imitating their language as much as he could. As a result, his pace was so slow, and the sun rose so quickly and ardently, that it would have melted his brains if he had had any.
……
During this time, Don Quixote approached a farmer who was a neighbor of his, a good man—if that title can be given to someone who is poor—but without much in the way of brains. In short, he told him so much, and persuaded and promised him so much, that the poor peasant resolved to go off with him and serve as his squire. Among other things, Don Quixote said that he should prepare to go with him gladly, because it might happen that one day he would have an adventure that would gain him, in the blink of an eye, an ínsula, and he would make him its governor. With these promises and others like them, Sancho Panza, for that was the farmer’s name, left his wife and children and agreed to be his neighbor’s squire.
……
As they were talking, they saw thirty or forty of the windmills found in that countryside, and as soon as Don Quixote caught sight of them, he said to his squire: “Good fortune is guiding our affairs better than we could have desired, for there you see, friend Sancho Panza, thirty or more enormous giants with whom I intend to do battle and whose lives I intend to take, and with the spoils we shall begin to grow rich, for this is righteous warfare, and it is a great service to God to remove so evil a breed from the face of the earth.”
“What giants?” said Sancho Panza.
“Those you see over there,” replied his master, “with the long arms; sometimes they are almost two leagues long.”
“Look, your grace,” Sancho responded, “those things that appear over there aren’t giants but windmills, and what looks like their arms are the sails that are turned by the wind and make the grindstone move.”
“It seems clear to me,” replied Don Quixote, “that thou art not well-versed in the matter of adventures: these are giants; and if thou art afraid, move aside and start to pray whilst I enter with them in fierce and unequal combat.”
And having said this, he spurred his horse, Rocinante, paying no attention to the shouts of his squire, Sancho, who warned him that, beyond any doubt, those things he was about to attack were windmills and not giants. But he was so convinced that they were giants that he did not hear the shouts of his squire, Sancho, and could not see, though he was very close, what they really were; instead, he charged and called out:
“Flee not, cowards and base creatures, for it is a single knight who attacks you.”
Just then a gust of wind began to blow, and the great sails began to move, and, seeing this, Don Quixote said:
“Even if you move more arms than the giant Briareus, you will answer to me.”
And saying this, and commending himself with all his heart to his lady Dulcinea, asking that she come to his aid at this critical moment, and well-protected by his shield, with his lance in its socket, he charged at Rocinante’s full gallop and attacked the first mill he came to; and as he thrust his lance into the sail, the wind moved it with so much force that it broke the lance into pieces and picked up the horse and the knight, who then dropped to the ground and were very badly battered. Sancho Panza hurried to help as fast as his donkey could carry him, and when he reached them he discovered that Don Quixote could not move because he had taken so hard a fall with Rocinante.
“God save me!” said Sancho. “Didn’t I tell your grace to watch what you were doing, that these were nothing but windmills, and only somebody whose head was full of them wouldn’t know that?”
“Be quiet, Sancho my friend,” replied Don Quixote. “Matters of war, more than any others, are subject to continual change; moreover, I think, and therefore it is true, that the same Frestón the Wise who stole my room and my books has turned these giants into windmills in order to deprive me of the glory of defeating them: such is the enmity he feels for me; but in the end, his evil arts will not prevail against the power of my virtuous sword.”
“God’s will be done,” replied Sancho Panza.
###
…
Second Part of the Ingenious Gentleman Don Quixote of La Mancha
By Miguel de Cervantes Saavedra Author of the First Part
Dedication
Dedicated to Don Pedro Fernández de Castro, Count of Lemos, Andrade, and Villalba, Marquis of Sarria, Gentleman-in-waiting to His Majesty, Commander of the jurisdiction of Peñafiel and La Zarza, member of the Order of Alcántara, Viceroy, Governor, and Captain General of the Kingdom of Naples, and President of the Supreme Council of Italy.
To the Count of Lemos
SOME DAYS AGO, when I sent Your Excellency my plays, printed before they were performed, I said, if I remember correctly, that Don Quixote had his spurs ready to make the journey to kiss Your Excellency’s hands, and now I say that he is wearing them, and is on his way, and if he arrives, it seems to me I will have performed a service for Your Excellency, because I have been urged on every side to send him forth in order to alleviate the loathing and disgust caused by another Don Quixote who has traveled the world in the disguise of a second part, and the person who has shown the deepest interest has been the great Emperor of China, who, not more than a month ago, sent an emissary with a letter for me in the Chinese language, asking, or I should say begging, me to send the knight to him, because he wanted to establish a college in which the Castilian language would be read, and the book he wanted the students to read was the history of Don Quixote. He further said that he wanted me to be the rector of the college.
I asked the bearer of the letter if His Majesty had given him anything that would help me to defray my expenses. He replied that it had not even occurred to him.
“Well, brother,” I responded, “you can go back to your China, covering your ten leagues a day, or twenty, or whatever you prefer; because my health is not good enough for me to undertake so long a journey, and not only am I ailing but I am lacking in funds, and emperor for emperor and monarch for monarch, in Naples I have the great Count of Lemos, who, without all the provisos of colleges and rectorships, sustains me and protects me and does me more good turns than I could ever desire.”
With this I took my leave of him, and with this I take my leave now, offering to Your Excellency The Travails of Persiles and Sigismunda, a book that I will complete in four months, Deo volente, and it will be either the worst or best ever composed in our language, I mean, of those written for diversion; I must say I regret having said the worst, because in the opinion of my friends it is bound to reach the extremes of possible goodness. May Your Excellency enjoy all the good health we wish for you; Persiles will soon be ready to kiss your hands, and I, your feet, being, as I am, the servant of Your Excellency. In Madrid, the last day of October, 1615.
Your Excellency’s servant,
MIGUEL DE CERVANTES SAAVEDRA
Prologue to the Reader
LORD SAVE ME, how impatiently you must be waiting for this prologue, illustrious or perhaps plebeian reader, believing you will find in it reprisals, quarrels, and vituperations hurled at the author of the second Don Quixote. I mean the one sired in Tordesillas, they say, and born in Tarragona!1 But the truth is I will not give you that pleasure, for although offenses awaken rage in the most humble hearts, in mine this rule must find its exception. You would like me to call him an ass, a fool, an insolent dolt, but the thought has not even entered my mind: let his sin be his punishment, let him eat it with his bread, and let that be an end to it. What I do mind, however, is that he accuses me of being old and one-handed, as if it had been in my power to stop time and halt its passage, or as if I had been wounded in some tavern and not at the greatest event ever seen in past or present times, or that future times can ever hope to see. If my wounds do not shine in the eyes of those who see them, they are, at least, esteemed by those who know where they were acquired; it seems better for a soldier to be dead in combat than safe in flight, and I believe this so firmly that even if I could achieve the impossible now, I would rather have taken part in that prodigious battle than to be free of wounds and not to have been there. The wounds on a soldier’s face and bosom are stars that guide others to the heaven of honor and the desire to win glory, and it should be noted that one writes not with gray hairs but with the understanding, which generally improves with the years.
I am also sorry that he calls me envious and, as if I were an ignorant man, explains to me what envy is, but the fact is that of the two kinds of envy, I know only the one that is holy, noble, and well-intentioned; and this being so, I do not need to persecute any priest, especially one who is a familiar of the Holy Office; if he said this in defense of the person on whose behalf he appears to have said it, he was entirely deceived, for I revere that person’s genius and admire his works and continual and virtuous diligence. But I do thank this worthy author for saying that although my novels are more satiric than exemplary, they are good, which they could not be if they were not good in every respect.
I think you will say that I am showing great restraint and am keeping well within the bounds of modesty, knowing that one must not add afflictions to the afflicted, and the affliction of this gentleman is undoubtedly very great, for he does not dare to appear openly in the light of day but hides his name and conceals his birthplace, as if he had committed some terrible act of treason against the crown. If you ever happen to meet him, tell him for me that I do not consider myself offended, for I know very well what the temptations of the devil are, and one of the greatest is to give a man the idea that he can compose and publish a book and thereby win as much fame as fortune, as much fortune as fame; in confirmation of this, I should like you, using all your wit and charm, to tell him this story:
In Sevilla there was a madman who had the strangest, most comical notion that any madman ever had. What he did was to make a tube out of a reed that he sharpened at one end, and then he would catch a dog on the street, or somewhere else, hold down one of its hind legs with his foot, lift the other with his hand, fit the tube into the right place, and blow until he had made the animal as round as a ball, and then, holding it up, he would give the dog two little pats on the belly and let it go, saying to the onlookers, and there were always a good number of them:
“Now do your graces think it’s an easy job to blow up a dog?
” Now does your grace think it’s an easy job to write a book?
And if that story does not please him, my dear reader, you can tell him this one, which is also about a madman and a dog:
In Córdoba there was a madman who was in the habit of carrying on his head a slab of marble, or a stone of no small weight, and when he came across an unwary dog, he would go up to the animal and drop the weight straight down on it. The dog would go into a panic and, barking and howling, run up three streets without stopping. Now, one of the dogs he dropped the weight on happened to belong to a haberdasher and was much loved by its owner. The stone came down, hit the dog on the head, and the battered animal began to yelp and howl; its master saw and heard this, and he seized a measuring stick, came after the madman, and beat him to within an inch of his life, and with each blow he said:
“You miserable thief, you dog, why did you hurt my hound? Didn’t you see, cruel man, that my dog was a hound?”
And repeating the word hound over and over again, he beat and pummeled the madman. Chastised, the madman withdrew and was not seen on the street for more than a month, but at the end of this time he returned with the same mad idea and an even heavier weight. He would go up to a dog, stare at it long and hard, and not wanting or daring to drop the stone, he would say:
“This is a hound: watch out!”
In fact, all the dogs he encountered, even if they were mastiffs or little lapdogs, he called hounds, and so he never dropped a stone on one again. Perhaps something similar may happen to this storyteller, who will not dare ever again to set his great talent loose among books, which, when they are bad, are harder than boulders.
Tell him, too, that his threat to deprive me of profits with his book is something I do not care about at all, for in the words of the famous interlude La Perendenga, I say long live my Lord High Mayor, and the peace of Christ be with you all. Long live the great Count of Lemos, whose well-known Christianity and liberality keep me standing in spite of all the blows struck by my bad fortune, and long live the supreme charity of His Eminence of Toledo, Don Bernardo de Sandoval y Rojas, even if there were no more printing presses in the world, and even if more books were published against me than there are letters in the verses of Mingo Revulgo. These two princes have not received adulation from me, or flattery, but moved only by their own goodness, they have undertaken to favor me with kindness, and for this I consider myself luckier and richer than if fortune had brought me to the heights by any ordinary means. A poor man may have honor, but not a villain; need may cloud nobility, but not hide it completely; if virtue sheds her light, even along the crags and cracks of poverty, it will be esteemed by high, noble spirits, and so be favored.
Say no more to him, and I do not wish to say more to you except to tell you to consider that this second part of Don Quixote, which I offer to you now, is cut by the same artisan and from the same cloth as the first, and in it I give you a somewhat expanded Don Quixote who is, at the end, dead and buried, so that no one will dare tell more tales about him, for the ones told in the past are enough, and it is also enough that an honorable man has recounted his clever follies and does not want to take them up again; for abundance, even of things that are good, makes people esteem them less, and scarcity, even of bad things, lends a certain value. I forgot to tell you to expect the Persiles, which I am finishing now, and the second part of Galatea.
Chapter I
Regarding what transpired when the priest and the barber discussed his illness with Don Quixote
Cide Hamete Benengeli tells us in the second part of this history, which recounts the third sally of Don Quixote, that the priest and the barber did not see the knight for almost a month in order not to restore and bring back to his mind events of the past, but this did not stop them from visiting his niece and housekeeper, charging them to be sure to pamper him and give him food to eat that would strengthen and fortify his heart and brain, the source, as they had good reason to think, of all his misfortunes. The two women said that they already were doing so, and would continue to do so, as willingly and carefully as possible, because they could see that there were moments when their lord and master gave signs of being in his right mind; this made the priest and the barber extremely happy, for then it seemed to them that they had done the right thing by bringing him home, enchanted, in the oxcart, as recounted in the final chapter of the first part of this great and accurate history. And so they decided to visit him and see his improvement for themselves, although they considered a complete cure almost impossible, and they agreed not to make any mention at all of knight errantry so as not to run the risk of reopening his wounds, which were still so fresh.
In short, they visited him and found him sitting up in bed, dressed in the green flannel vest he wore under his armor, and a red Toledan cap, and looking so dry and gaunt that he seemed to be a mummy. They received a warm welcome, they asked after his health, and he accounted for himself and the state of his health with very good judgment and in very elegant words, and in the course of their conversation they began to discuss what is called reason of state and ways of governing, correcting this abuse and condemning that one, reforming one custom and eliminating another, each one of the three becoming a new legislator, a modern Lycurgus, a latter-day Solon,1 and they so transformed the nation that it seemed as if they had placed it in the forge and taken out a new one, and Don Quixote spoke with so much intelligence regarding all the subjects they touched upon that his two examiners thought there was no doubt that he was completely well and his sanity restored.
The niece and housekeeper were present at this conversation, and they never tired of giving thanks to God at seeing their lord and master with all his wits; the priest, however, changing his earlier intention, which was not to touch on chivalric matters, wanted a more thorough test of whether or not Don Quixote’s recovery was false or true, and so he gradually began to recount news of the court, and among other things, he said it was thought certain that the Turk would come down with a powerful fleet, but no one knew his plans or where the huge cloud would burst; this fear, which has us on the alert almost every year, had now affected all of Christendom, and His Majesty had fortified the coasts of Naples and Sicily and the island of Malta. To which Don Quixote responded:
“His Majesty has behaved like a most prudent warrior by fortifying his states in good time so that the enemy will not find them unprepared, but if he were to take my advice, I would counsel him to take a precautionary measure that His Majesty is very far from considering at present.”
As soon as the priest heard this, he said to himself:
“May God hold you in his hand, my poor Don Quixote, for it seems to me you have leaped from the high peak of your madness into the profound abyss of your foolishness!”
But the barber, who had already had the same thought as the priest, asked Don Quixote to tell him the precautionary measure he thought it would be good to undertake; perhaps it might be put on the list of the many impertinent proposals that are commonly offered to princes.
“Mine, Señor Shaver,” said Don Quixote, “is not impertinent but completely pertinent.”
“I don’t say it isn’t,” replied the barber, “but experience shows that all or most of the schemes presented to His Majesty are either impossible, or absurd, or harmful to the king and his kingdom.”
“Well, mine,” responded Don Quixote, “is neither impossible nor absurd, but is, rather, the easiest, most just, most practical, and shrewdest that has ever occurred to any planner.”
“Your grace is slow in telling us what it is, Señor Don Quixote,” said the priest.
“I would not want,” said Don Quixote, “to state it here and now, and tomorrow have it find its way to the ears of the king’s advisers so that another receives the thanks and rewards for my labor.”
“As far as I am concerned,” said the barber, “I give my word, here and before God, not to repeat what your grace would tell the king or the rook or any man on earth, a vow I learned in the tale of the priest who, in the preface, told the king about the thief who had stolen one hundred doblas from him, as well as his mule with the ambling gait.”
“I know nothing of stories,” said Don Quixote, “but I do know this is a good vow because I know the barber is a trustworthy man.”
“Even if he were not,” said the priest, “I would vouch for him and guarantee that in this case he will say no more than if he were mute, under pain of sentence by the court.”
“And who vouches for your grace, Señor Priest?” said Don Quixote.
“My profession,” responded the priest, “which is to keep secrets.”
“By my faith!” Don Quixote said then. “What else can His Majesty do but command by public proclamation that on a specific day all the knights errant wandering through Spain are to gather at court, and even if no more than half a dozen were to come, there might be one among them who could, by himself, destroy all the power of the Turk. Your graces should listen carefully and follow what I say. Is it by any chance surprising for a single knight errant to vanquish an army of two hundred thousand men, as if all of them together had but one throat or were made of sugar candy? Tell me, then: how many histories are filled with such marvels? If only—to my misfortune, if not to anyone else’s—the famous Don Belianís were alive today, or any one of the countless descendants of Amadís of Gaul! If any of them were here today and confronted the Turk, it would not be to his advantage! But God will look after His people and provide one who, if not as excellent as the knights errant of old, at least will not be inferior to them in courage; God understands me, and I shall say no more.”
“Oh!” said the niece at this point. “You can kill me if my uncle doesn’t want to be a knight errant again!”
To which Don Quixote said:
“I shall die a knight errant, and let the Turk come down or go up whenever he wishes and however powerfully he can; once again I say that God understands me.”
And then the barber said:
“I beg your graces to give me leave to tell a brief story that occurred in Sevilla; since it is very much to the point here, I should like to tell it now.”
Don Quixote gave his permission, and the priest and the others listened carefully, and the barber began in this manner:
“In the madhouse in Sevilla was a man whose relatives had put him there because he had lost his reason. He was a graduate in canon law from Osuna, but even if he had graduated from Salamanca, in the opinion of many he would not have been any less mad. This graduate, after some years of confinement, came to believe that he was sane and in his right mind, and with this thought he wrote to the archbishop earnestly entreating him, in carefully chosen phrases, to have him removed from the misery in which he was living, for through God’s mercy he had now recovered his reason, but his relatives, in order to enjoy his share of the estate, were keeping him there, and despite the truth would have wanted him mad until his death.
The archbishop, persuaded by his many well-written and well-reasoned letters, ordered one of his chaplains to learn from the superintendent of the madhouse if what the licentiate had written was true, and to speak to the madman as well, and, if it seemed he was in his right mind, to release him and set him free. The chaplain did so, and the superintendent told him that the man was still mad; though he often spoke like a person of great intelligence, he eventually would begin to say countless foolish things, as many and as deeply felt as his earlier rational statements, and this the chaplain could see for himself if he spoke to him. The chaplain agreed, visited the madman, spoke to him for more than an hour, and in all that time the madman never made a confused or foolish statement; rather, he spoke so judiciously that the chaplain was obliged to believe that the madman was sane; one of the things the madman told him was that the superintendent bore him ill will because he did not want to lose the gifts his relatives gave him for saying he was still mad, though with periods of lucidity; the greatest obstacle for him in his misfortune was his wealth, because in order to enjoy it, his enemies were deceptive and denied the mercy Our Lord had shown by turning him from a beast back into a man. In short, what he said depicted the superintendent as suspect, his relatives as greedy and heartless, and himself as so reasonable that the chaplain resolved to take him back so that the archbishop could see and touch the truth of the matter for himself. …
###







































































































































































































































































































































































































































































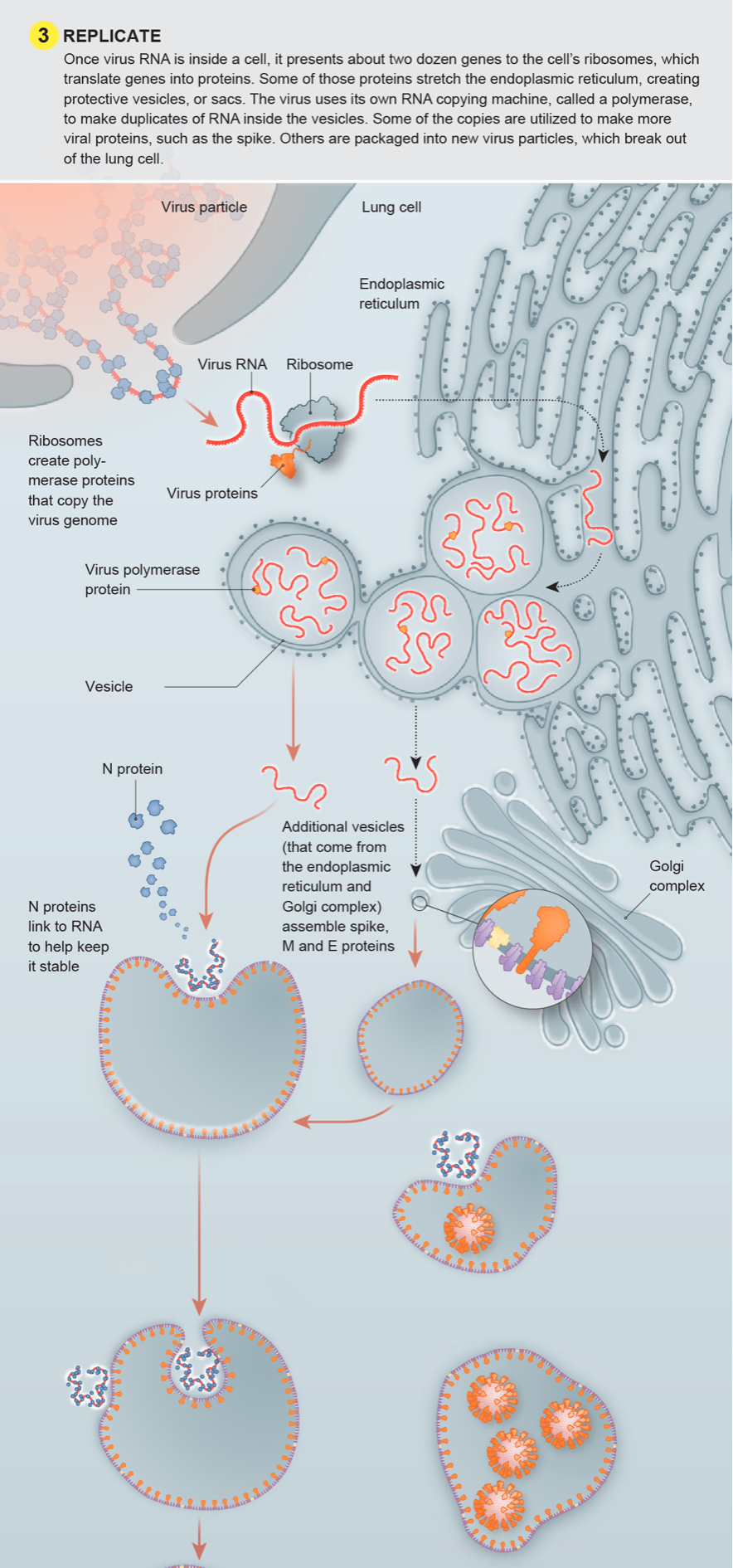






















































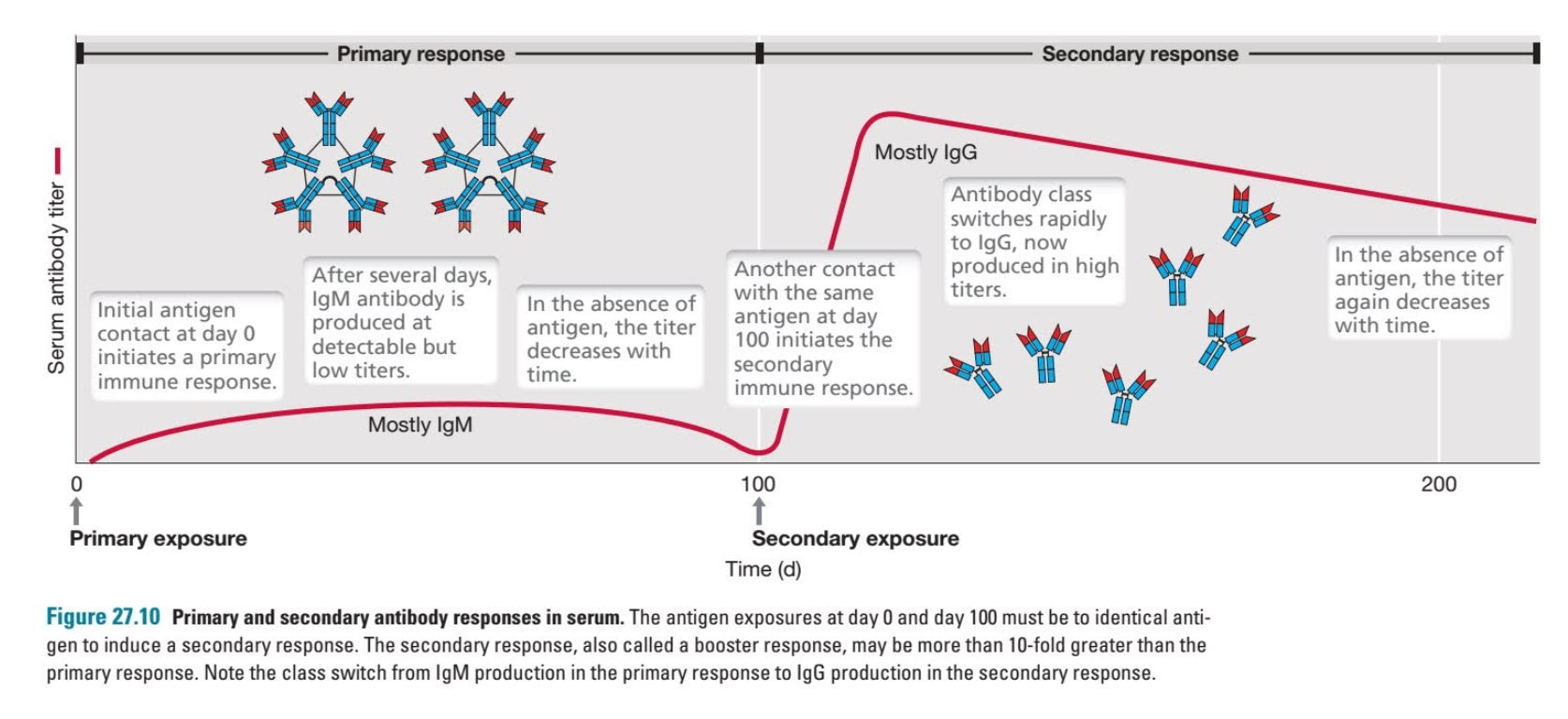











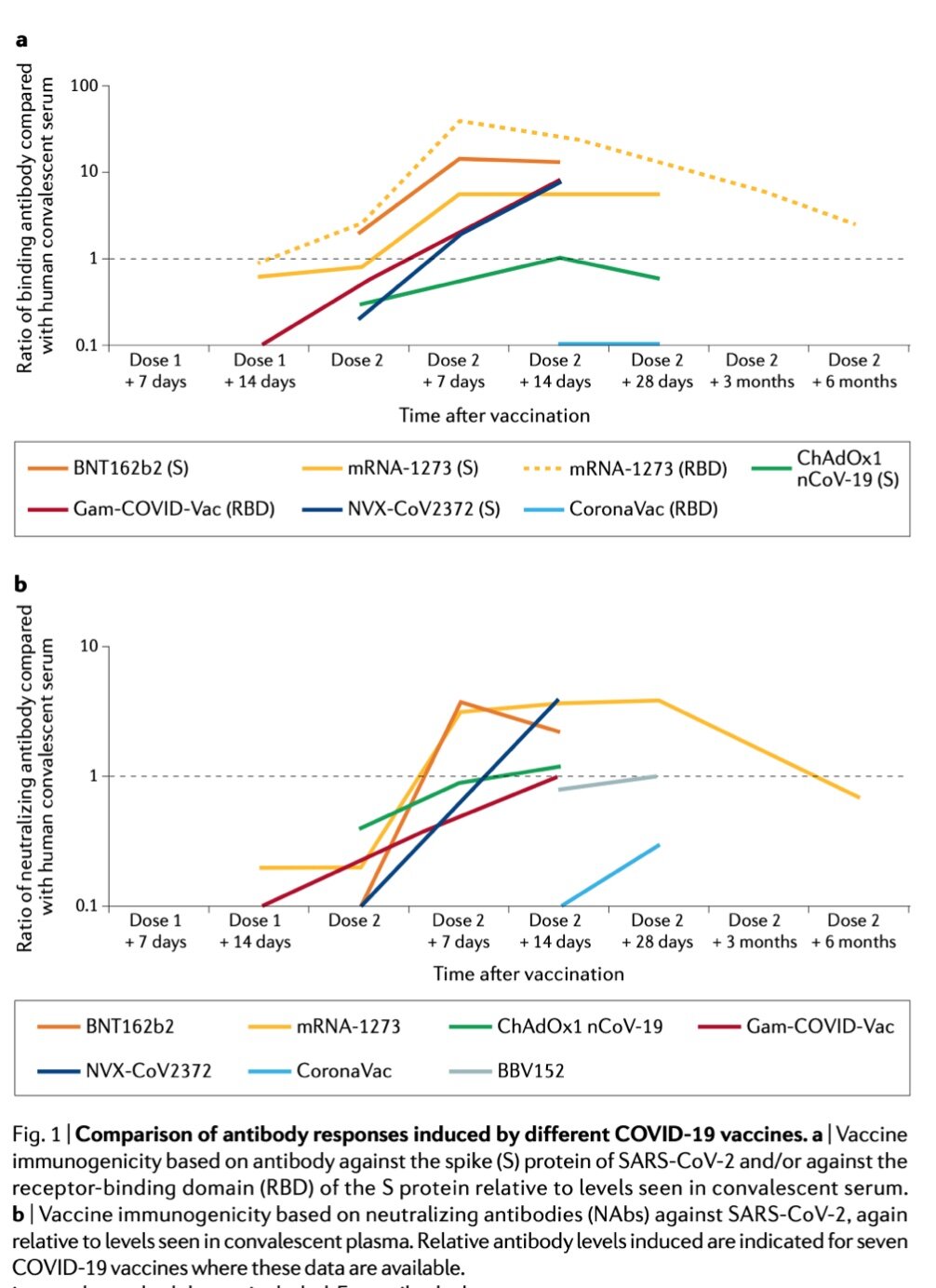






























![book info ( … for 7th edition - 2022 ) excerpts: All channels and many transporters [in the membrane] allow solutes to cross the membrane only passively (“downhill”), a process called passive transport. In the case of transport of](https://images.squarespace-cdn.com/content/v1/522b22f2e4b04879e6b51037/1673744329732-A0RLBWOBOF1T2YDOQZXU/Screenshot+2023-01-14+at+6.47.37+PM.png)















































































































!["From the abundance, accessibility and richness of this iron ore, its proximity to the coal field and to the navigation of the Tombigbee River, I can hardly doubt that, like the coal itself, [Birmingham] is destined, at no distant day, to be a](https://images.squarespace-cdn.com/content/v1/522b22f2e4b04879e6b51037/1542471845701-GA3WE46HVGOFINB6B01T/Screen+Shot+2018-11-17+at+10.20.19+AM.png)